Flucinar 15g is formulated as an ointment commonly used to treat various skin diseases. It is important to be aware of the relevant information about the medication to use it correctly and avoid any potential side effects. To better understand Flucinar and its uses, please refer to the detailed information in the article below.
1. What is Flucinar?
Flucinar contains Fluocinolone as the active ingredient, which belongs to the group of topical antifungal and antiparasitic medications. The medication is formulated as an ointment for external use, with a concentration of 15 grams, and is manufactured by Pharmaceuticals Works Jelfa S.A. in Poland.
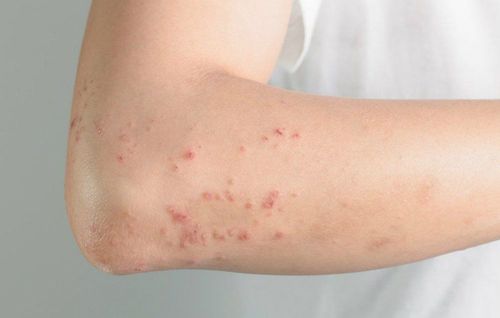
It is commonly used in the treatment of symptoms of dermatological diseases, including:
- Allergic dermatitis, seborrheic dermatitis.
- Erythema multiforme, urticaria related to lichen disease, chronic psoriasis.
- Dry, non-infected dermatitis responsive to glucocorticosteroids, accompanied by keratosis or persistent itching symptoms.
- Systemic lupus erythematosus, allergic contact eczema, lichen planus.
2. Uses of Flucinar 15g
2.1. The pharmacodynamics of Flucinar 15g
The active ingredient, Fluocinolone acetonide, is a synthetic corticosteroid with low to moderate potency, featuring a fluorine atom attached to the steroid nucleus. The mechanism of action of a topical corticosteroid is due to the combination of three key properties: anti-pruritic, anti-inflammatory, and vasoconstrictive effects.
Corticosteroids reduce the release, formation, and limit the effects of vasoconstrictor substances released during inflammation (such as histamines, kinins, lysosomal enzymes, leukotrienes, prostaglandins, and complement components), thereby helping to reduce inflammation. Steroids also reduce cell membrane permeability. Through vasoconstriction, fluocinolone helps to decrease fluid leakage in inflamed skin areas. The medication increases collagen accumulation and protein synthesis, slows down protein proliferative processes, and promotes protein degradation in the skin.
Corticosteroids, particularly those containing fluorine, counteract the mitotic activity of the upper epidermal cells. Repeated use of fluorine-containing corticosteroids reduces the drug's effects on the skin's surface, leading to a prolonged time for the drug to take effect, but also increasing unwanted side effects and systemic absorption.
2.2. The pharmacokinetics of Flucinar 15g
- Absorption:
When applied topically to intact skin, only a small amount of the corticosteroid reaches the dermis and subsequently enters the systemic circulation. However, absorption increases significantly when the skin loses its keratin layer, such as in cases of inflammation and/or other diseases affecting the epidermal barrier (eczema, psoriasis). The absorption depends on the degree of skin penetration, the thickness of the medication applied to the skin and the condition of the skin at the application site. Specifically, the drug is absorbed more in areas such as the eyelids (about 40%) and scrotum (about 36%) compared to the forehead (about 7%), scalp (about 4%), or forearm (about 1%). It is also more absorbed in areas with skin folds. The active ingredient, Fluocinolone acetonide, easily penetrates the stratum corneum of the skin at the site of application, and accumulation of the drug can be found at the site after 15 days of use.
- Metabolism:
Fluocinolone acetonide in Flucinar ointment is not metabolized in the skin. When the medication is applied according to the recommended instructions, a small amount of the drug is absorbed into the systemic circulation and is metabolized in the liver into inactive substances.
- Elimination:
The drug is primarily excreted via the kidneys in the form of glucuronides and sulfates, as well as in its unconjugated form. A small amount of metabolites is eliminated through feces.
2.3. Contraindications of Flucinar 15g
Flucinar 15g should not be used in the following cases:
- Hypersensitivity to any component of the drug.
- Bacterial, fungal, or viral skin infections.
- Seborrheic folliculitis, perioral dermatitis, rosacea.
- After vaccination.
- Children under 2 years old.
- Use with caution in patients with subcutaneous tissue atrophy, especially the elderly.
2.4. Side effects of Flucinar 15g
- On the skin
- Acne or pimples.
- Pus accumulation.
- Irritation, itching, blisters, crusting, or redness of the skin.
- Itching, pain, and burning sensation in areas with hair or pus at the hair follicles.
- Skin itching with small red blisters, resembling pinpricks.
- Dry, cracked skin.
- Skin darkening.
- Severe redness, itching, crusting, pain, or swelling of the skin.
- Swelling, itching, raised bumps on the skin surface, round, smooth, skin-colored, found in specific areas of the body.
- Small, raised, dark red spots resembling corn, especially when the medication is applied to the face.
Hearing: Changes in hearing, ear pain, or discomfort in the ear.
Vision: Using Flucinar ointment on the eyelid area may sometimes cause cataracts or glaucoma.
Systemic: Cushing's syndrome, suppression of the hypothalamic-pituitary-adrenal axis, hypertension, hyperglycemia, edema, and immune suppression.
2.5. Precautions for using Flucinar 15g
- If you notice any signs of skin irritation or allergic reactions after applying Flucinar ointment, discontinue use of the medication.
- Avoid continuous use of the medication for more than 14 days. Prolonged use of the medication on large areas of skin may increase the frequency of side effects, causing edema, hyperglycemia (increased blood glucose levels), hypertension, and immune suppression.
- The systemic absorption of topical corticosteroids has been shown to cause reversible suppression of the hypothalamic-pituitary-adrenal (HPA) axis, resulting in symptoms such as hyperglycemia, Cushing's syndrome, and glucosuria in some patients.
- Conditions that increase the potential for systemic absorption include the use of stronger steroids, prolonged use, application to large surface areas, and occlusive dressings.
- Therefore, patients using a high dose of topical steroids on large surface areas or with occlusive dressings should be regularly evaluated for evidence of HPA axis suppression through tests such as urinary cortisol and free ACTH stimulation. If HPA axis suppression is detected, efforts should be made to reduce the medication, decrease the frequency of use, or switch to a lower-potency steroid.
- Recovery of HPA axis function generally occurs rapidly and is complete after discontinuing the medication. In cases of increased infection at the site of application, appropriate antifungal and antibacterial treatments should be initiated. If infection symptoms persist, discontinue the use of the ointment until the infection resolves.
- Caution to prevent Flucinar ointment from coming into contact with the eyes, mucous membranes, or open wounds.
- Avoid using the medication on the eyelids or near the eyes in patients with wide-angle or narrow-angle glaucoma, and in patients with cataracts, as it may worsen the clinical symptoms of these conditions.
- Use with particular caution in patients with psoriasis, as the use of topical corticosteroids can be dangerous for several reasons, including disease relapse due to increased drug tolerance in patients with pustular psoriasis and systemic toxicity due to skin integrity loss.
- Use the medication on the face, armpits, and groin only when absolutely necessary, as absorption is increased and unwanted side effects (capillary dilation, perioral dermatitis) may occur even after short-term use.
- Use cautiously in cases of subcutaneous atrophy, particularly in elderly patients.
- Flucinar ointment contains propylene glycol, which may cause allergic reactions. Therefore, patients with a history of allergy to propylene glycol should consult with a doctor before using this medication.
- Patients should not cover or wrap the affected skin area after applying the medication unless instructed to do so by a doctor. Occluding large skin areas while using the medication may increase the risk of systemic toxicity.
- Pregnant and breastfeeding women should avoid using this medication unless prescribed by a doctor.
- Pediatric and Adolescent: Children may have an increased risk of hypothalamic-pituitary-adrenal (HPA) axis disorders and Cushing's syndrome due to the systemic effects of corticosteroids. Children's skin surface area is larger relative to body mass. Long-term corticosteroid treatment can also cause growth and development disturbances in children.
2.6. Drug interactions
No interactions have been reported with the use of topical medication. However, the varicella vaccine and other immunosuppressive drugs should not be administered, as they can reduce the broad immune response, particularly when the medication is applied over large areas of the skin.
To ensure safety and efficacy, inform your doctor about all medications, health supplements, and any other diseases you may have to prevent any adverse interactions.
3. Effective Use of Flucinar 15g
Proper Use of Flucinar:
- Wash your hands with soap and water before and after applying fluocinolone.
- It is crucial to use fluocinolone exactly as prescribed by your doctor. Do not apply a thick layer, use it more frequently, or extend the treatment duration beyond the prescribed period. Doing so may lead to unwanted side effects or skin irritation.
- The dosage should be adjusted based on the severity and nature of each condition, as well as the individual patient’s sensitivity. Fluocinolone should not be used to treat certain infections or skin conditions, such as severe burns.
- Apply only a small amount of the medication to the affected area, avoiding spreading it to surrounding skin. Do not apply more than 1–2 times per day.
- Do not cover the treated area with a bandage unless instructed by a doctor. However, for psoriasis, occlusion therapy may be necessary, with daily dressing changes.
- Use on the facial skin: Not more than 7 days.
- Do not use more than 1 tube of Flucinar 15g ointment per week.
- Children and adolescents: Do not use the medication for children under 2 years old.
- Caution when using the medication for children over 2 years old. Use the medication only when absolutely necessary, once a day, and do not apply it on the child's face. If the medication is used on the diaper area of infants, avoid using diapers or tight clothing unless directed by a doctor.
To arrange an appointment, please call HOTLINE or make your reservation directly HERE. You may also download the MyVinmec app to schedule appointments faster and manage your reservations more conveniently.